Preterm infants with BPD
Why change is needed
Preterm infants with immature lungs are among the most vulnerable patients in medicine. Around one in ten babies is born preterm. Many require respiratory support in neonatal intensive care and face a high risk of developing bronchopulmonary dysplasia (BPD), a chronic lung disease associated with long hospital stays, high ICU resource use, and long-term respiratory impairment.
Inhaled therapies are clinically attractive because they can act directly in the lungs while reducing systemic exposure. However, in neonatal care this approach is still technically limited. Under respiratory support or mechanical ventilation, conventional aerosol delivery leads to major drug losses in the breathing circuit. As a result, lung deposition is low, inconsistent, and clinically difficult to rely on.
Clinicians are therefore often forced into an unfavorable trade-off: systemic medication with relevant whole-body exposure. In preterm infants, this comes with clinically meaningful side effects and the risk of long-term harm, which is why it is typically used only when no effective local alternative exists. This is the core care gap InhalPlus is addressing.
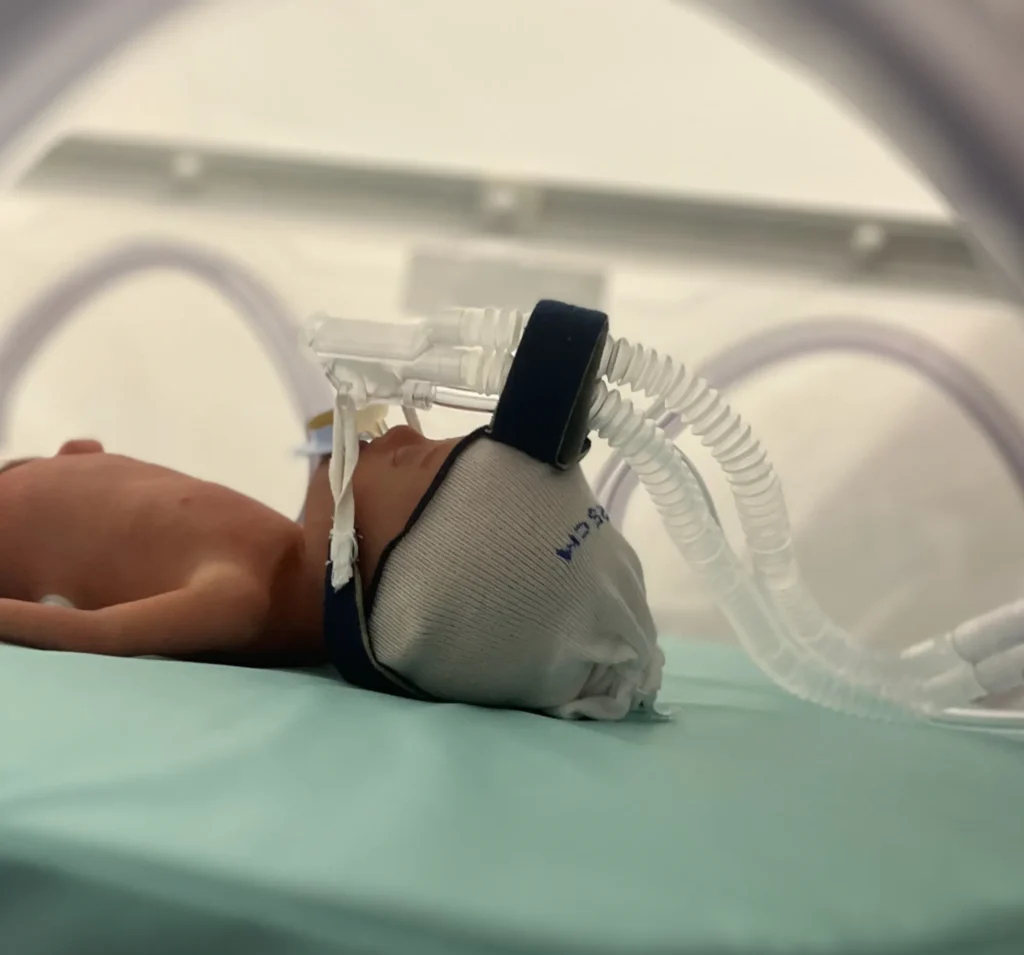
InhalPlus closes this gap with a patented, non-invasive bypass technology for aerosol drug delivery in neonatology.
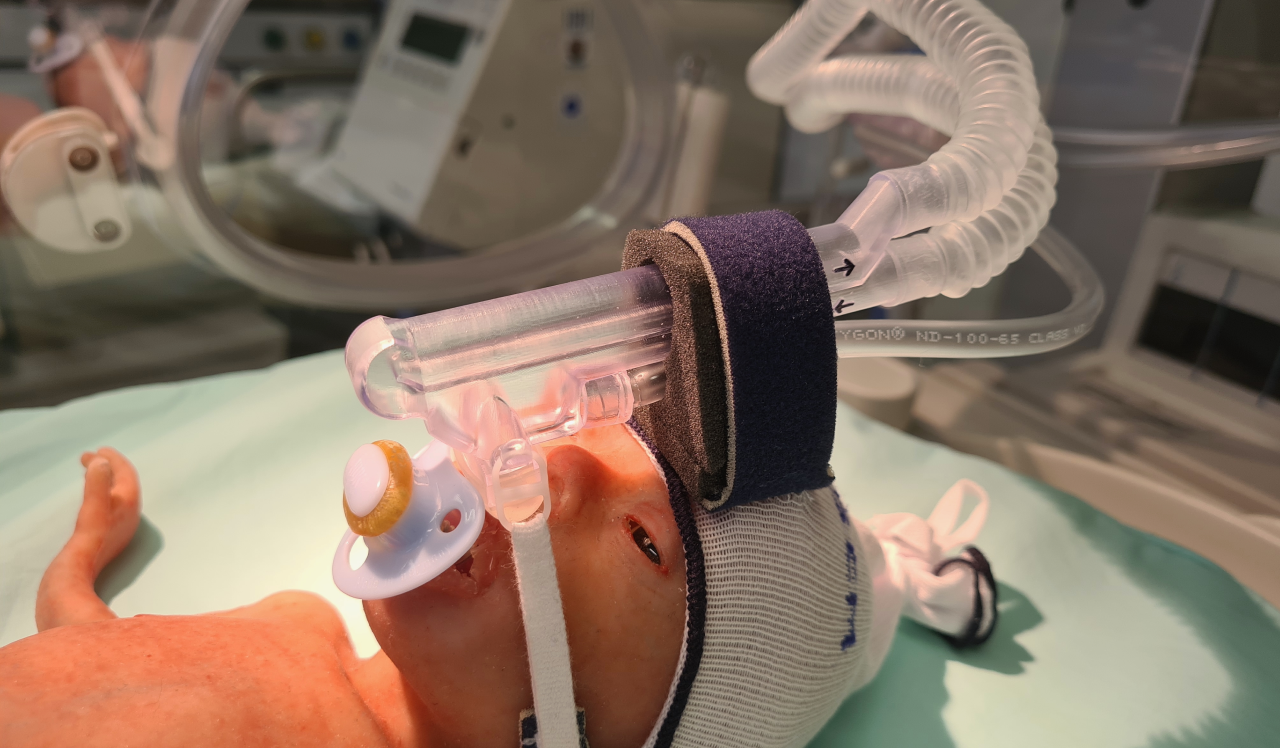
InhalPlus closes this gap with a patented, non-invasive bypass technology for aerosol drug delivery in neonatology.

Our Solution
Instead of introducing aerosol into the main flow of the breathing circuit (where it becomes diluted and lost), our system bypasses the dilution effect and delivers a highly concentrated aerosol directly to the patient’s airways. This enables highly efficient and reproducible pulmonary deposition while respiratory support or mechanical ventilation continues uninterrupted.
The system is compatible with standard ventilators and mesh nebulizers and integrates seamlessly into existing NICU workflows.
Our initial focus is inhaled therapy for high-risk preterm infants to prevent or mitigate BPD.
By enabling effective local therapy without invasive escalation, InhalPlus has the potential to stabilize respiratory courses, reduce complications, and improve long-term outcomes.
Market potential
The market is highly defined and clinically anchored: preterm infants treated under respiratory support in specialized NICUs, particularly those with prolonged courses and high BPD risk.
Globally, around 15 million infants are born prematurely each year. In Germany and Switzerland alone, the target population includes approximately 34,000 preterm infants per year receiving CPAP therapy, including the high-risk group most affected by chronic respiratory complications. Extrapolated to the global market, this corresponds to an estimated target population of around 8.7 million preterm infants every year.
Neonatal intensive care is structurally centralized. A relatively small number of top-level NICUs treat a large share of the relevant patient population. This enables a focused market entry, rapid reference building, and efficient scaling across key centers.
From a health-economic perspective, the burden of BPD is substantial. Infants with chronic respiratory complications often require long ICU stays and sustained high-intensity care. Even moderate reductions in length of stay, escalation events, or long-term morbidity can generate significant value for hospitals and healthcare systems.